Live Better
Reaching your personal goals with a chronic illness

(BPT) – Content sponsored and provided by Pfizer.
Janet H., a wife and mother from Alabama, had many things to celebrate in her life – two beautiful children, a wonderful husband, her dog Falco, and a rewarding career. She was proud that even with all of this, she was able to achieve her personal goal of staying active. However, she was diagnosed with a chronic disease the week before her 40th birthday.
Janet enjoyed moderate exercise, so when she began to have persistent joint pain and swelling in her knees, she suspected it was from an old injury. After initial visits with an orthopedic specialist, she was referred to a rheumatologist who diagnosed her with moderate to severe rheumatoid arthritis (RA) – a chronic, autoimmune disease of the joints.[i]
“I remember thinking that if this is what 40 is like, I don’t know if I can handle 50,” she says. “I wasn’t sure how RA would affect my life.”
Unfortunately, this story is all too common, as RA impacts approximately 1.6 million adults in the United States.[ii],[iii] Many living with a chronic condition like RA can feel that their life is put on hold while trying to manage their symptoms, doctor’s appointments, and the daily demands of life. Working toward personal goals can be put on the backburner, but does it have to be?
With her rheumatologist, Janet was motivated to find a treatment plan that could help relieve her RA symptoms before being prescribed XELJANZ® (tofacitinib) 5 mg twice daily tablets, an oral medication for adults with moderate to severe RA in which methotrexate did not work well enough. Janet’s rheumatologist reviewed the potential side effects of XELJANZ and explained that it is taken twice daily and has a BOXED WARNING for serious infections and malignancies.
Within three to six months, Janet felt improvement in her RA symptoms and found herself thinking about ways to become more active. She spoke with her rheumatologist and, together, they came up with exercise goals including yoga classes, cycling, and walking her dog with her husband and children. She also says one of her biggest milestones was walking her daughter down the aisle and gaining a new son-in-law. Eventually, Janet even worked with her rheumatologist to switch to a once-a-day version of XELJANZ, called XELJANZ XR extended release tablets.
Here are some of Janet’s tips to help someone living with a chronic illness work toward their personal goals:
Plan ahead: Living with a chronic condition can put a pause on spontaneity. Plan ahead when it comes to things like travel or big life events to allow ample time for adjustments and preparation.
Partner up: Find a significant other, friend or sibling who can join you on your journey toward reaching your goals. Sometimes living with a chronic condition requires extra support from others, so having a partner to plan with can help make that support seamless
Consult your doctor: Before considering any new travel or physical activity, make sure to consult your doctor. Discuss your plans and how you and your doctor can work together to help ensure you can meet these goals.By following these tips, Janet says that she has been able to better manage her RA and have more time to focus on her family, friends and herself.
“I hope my story can help other adults who are living with moderate to severe rheumatoid arthritis and inspire them to share their stories,” she says.
What is XELJANZ/XELJANZ XR?
XELJANZ/XELJANZ XR is a prescription medicine called a Janus kinase (JAK) inhibitor. XELJANZ/XELJANZ XR is used to treat adults with moderately to severely active rheumatoid arthritis in which methotrexate did not work well.
It is not known if XELJANZ/XELJANZ XR is safe and effective in people with hepatitis B or C.
XELJANZ/XELJANZ XR is not recommended for people with severe liver problems.
It is not known if XELJANZ/XELJANZ XR is safe and effective in children.
IMPORTANT SAFETY INFORMATION
What is the most important information I should know about XELJANZ/XELJANZ XR?
XELJANZ/XELJANZ XR may cause serious side effects, including:
Serious infections. XELJANZ/XELJANZ XR can lower the ability of your immune system to fight infections. Some people can have serious infections while taking XELJANZ/XELJANZ XR, including tuberculosis (TB), and infections caused by bacteria, fungi, or viruses that can spread throughout the body. Some people have died from these infections. Your healthcare provider should test you for TB before starting and during XELJANZ/XELJANZ XR treatment, and monitor you closely for signs and symptoms of TB infection during treatment. You should not start taking XELJANZ/XELJANZ XR if you have any kind of infection unless your healthcare provider tells you it is okay.
You may be at a higher risk of developing shingles (herpes zoster).
Before starting XELJANZ/XELJANZ XR, tell your healthcare provider if you:
think you have an infection or have symptoms of an infection, such as fever, sweating, or chills; cough; blood in phlegm; warm, red, or painful skin or sores on your body; burning when you urinate or urinating more often than normal; muscle aches; shortness of breath; weight loss; diarrhea or stomach pain; or feeling very tiredare being treated for an infectionget a lot of infections or have infections that keep coming backhave diabetes, chronic lung disease, HIV, or a weak immune system. People with these conditions have a higher chance for infectionshave TB, or have been in close contact with someone with TBlive or have lived in, or have traveled to certain parts of the country (such as the Ohio and Mississippi River valleys and the Southwest) where there is an increased chance for getting certain kinds of fungal infections (histoplasmosis, coccidioidomycosis, or blastomycosis). These infections may happen or become more severe if you use XELJANZ/XELJANZ XR. Ask your healthcare provider if you do not know if you have lived in an area where these infections are commonhave or have had hepatitis B or CAfter starting XELJANZ/XELJANZ XR, call your healthcare provider right away if you have any symptoms of an infection. XELJANZ/XELJANZ XR can make you more likely to get infections or make worse any infection that you have.
Cancer and immune system problems. XELJANZ/XELJANZ XR may increase your risk of certain cancers by changing the way your immune system works. Lymphoma and other cancers, including skin cancers, have happened in patients taking XELJANZ/XELJANZ XR. Tell your healthcare provider if you have ever had any type of cancer.
Some people who have taken XELJANZ with certain other medicines to prevent kidney transplant rejection have had a problem with certain white blood cells growing out of control (Epstein Barr Virus-associated post-transplant lymphoproliferative disorder).
Tears (perforation) in the stomach or intestines. Some people taking XELJANZ/XELJANZ XR can get tears in their stomach or intestine. This happens most often in people who also take nonsteroidal anti-inflammatory drugs (NSAIDs), corticosteroids, or methotrexate.
Tell your healthcare provider right away if you have fever and stomach-area pain that does not go away and a change in your bowel habits.
Changes in certain lab test results. Your healthcare provider should do blood tests before you start receiving XELJANZ/XELJANZ XR, and while you take XELJANZ/XELJANZ XR, to check for the following side effects:
changes in lymphocyte counts. Lymphocytes are white blood cells that help the body fight off infections.low neutrophil counts. Neutrophils are white blood cells that help the body fight off infections.low red blood cell count. This may mean that you have anemia, which may make you feel weak and tired.Your healthcare provider should routinely check certain liver tests.
You should not receive XELJANZ/XELJANZ XR if your lymphocyte count, neutrophil count, or red blood cell count is too low or your liver tests are too high. Your healthcare provider may stop your XELJANZ/XELJANZ XR treatment for a period of time if needed because of changes in these blood test results.
Your healthcare provider should do blood tests to check your cholesterol levels 4-8 weeks after you start XELJANZ/XELJANZ XR, and as needed after that.
What should I tell my healthcare provider before taking XELJANZ/XELJANZ XR?
Before taking XELJANZ/XELJANZ XR, tell your healthcare provider about all of your medical conditions, including if you:
have an infectionhave liver problemshave kidney problemshave any stomach area (abdominal) pain or been diagnosed with diverticulitis (inflammation in parts of the large intestine) or ulcers in your stomach or intestines, or narrowing within your digestive tracthave had a reaction to tofacitinib or any of the ingredients in XELJANZ/XELJANZ XRhave recently received or are scheduled to receive a vaccine. People taking XELJANZ/XELJANZ XR should not receive live vaccines but can receive non-live vaccinesplan to become pregnant or are pregnant. It is not known if XELJANZ/XELJANZ XR will harm an unborn baby. You should use effective birth control while you are taking XELJANZ/XELJANZ XR and for at least 4 weeks after you take your last dose.Pregnancy Registry: Pfizer has a registry for pregnant women who take XELJANZ/XELJANZ XR. The purpose of this registry is to check the health of the pregnant mother and her baby. If you are pregnant or become pregnant while taking XELJANZ/XELJANZ XR, talk to your healthcare provider about how you can join this pregnancy registry or you may contact the registry at 1-877-311-8972 to enrollplan to breastfeed or are breastfeedingTell your healthcare provider about all of the medicines you take, especially any other medicines to treat your rheumatoid arthritis. You should not take tocilizumab (Actemra®), etanercept (Enbrel®), adalimumab (Humira®), infliximab (Remicade®), rituximab (Rituxan®), abatacept (Orencia®), anakinra (Kineret®), certolizumab pegol (Cimzia®), golimumab (Simponi®), ustekinumab (Stelara®), secukinumab (Cosentyx®), azathioprine, cyclosporine, or other immunosuppressive drugs while you are taking XELJANZ/XELJANZ XR. Taking XELJANZ or XELJANZ XR with these medicines may increase your risk of infection.
Tell your healthcare provider if you are taking medicines that affect the way certain liver enzymes work. Ask your healthcare provider if you are not sure if your medicine is one of these.Taking XELJANZ/XELJANZ XR
When you take XELJANZ XR, you may see something in your stool that looks like a tablet. This is the empty shell from the tablet after the medicine has been absorbed by your body.
What are other possible side effects of XELJANZ/XELJANZ XR?
XELJANZ/XELJANZ XR may cause serious side effects, including hepatitis B or C activation infection in people who carry the virus in their blood. If you are a carrier of the hepatitis B or C virus (viruses that affect the liver), the virus may become active while you use XELJANZ/XELJANZ XR. Tell your healthcare provider if you have the following symptoms of a possible hepatitis B or C infection: feel very tired, little or no appetite, clay-colored bowel movements, chills, muscle aches, skin rash, skin or eyes look yellow, vomiting, fevers, stomach discomfort, or dark urine.
Common side effects of XELJANZ/XELJANZ XR include upper respiratory tract infections (common cold, sinus infections), headache, diarrhea, and nasal congestion, sore throat, and runny nose (nasopharyngitis).
To learn more about XELJANZ/XELJANZ XR, a treatment option for moderately to severely active RA, visit XELJANZ.com.
Please click the direct link to the full US Prescribing Information for XELJANZ/XELJANZ XR, including BOXED WARNING and Medication Guide: http://labeling.pfizer.com/ShowLabeling.aspx?id=959.
[i] National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS). National Institutes of Health, Department of Health and Human Services. Handout on health: rheumatoid arthritis. August 2014. http://www.niams.nih.gov/Health_Info/Rheumatic_Disease/default.asp. Accessed December 4, 2017.
[ii] Sacks J, Lou Y, Helmick, C. Prevalence of specific types of arthritis and other rheumatic conditions in the ambulatory health care system in the United States 2001-2005. Arthritis Care Res. 2010;62(4):460-464.
[iii] Howden L, Meyer J. 2010 U.S. Census Bureau results – U.S. Census Bureau, 2010 Census Summary File 1.
Live Better
Steps to get your yard ready for spring

(Family Features) As the weather begins to warm and days get longer, it’s important to begin preparing your yard for a healthy growing season. While factors like climate, soil type and grass type can all impact how your yard grows, there are a few steps you can take during the spring months to help your lawn thrive.
From dethatching and aerating to fertilizing and mowing, tackling these simple chores can help ensure your yard looks its best and is prepared to fight off seasonal weeds, disease and drought.
Tune Your Mower
Before it’s time for the first mowing of the season, inspect your mower and perform any routine maintenance necessary. That goes beyond sharpening, or replacing, the blade, and includes changing the oil, spark plugs and filter as well as filling with a fresh tank of gas.
Loosen the Thatch Layer
While it’s important to avoid working on your lawn until after the final freeze to avoid damaging the grass, raking your yard with a spring tine rake to loosen thatch – the layer of leaves, roots and dead grass that builds up between live grass and soil – before the first mow is equally important. Be sure to rake when the soil is dry; if it’s too soft or muddy, you may pull up healthy grass crowns.
Combat Compacted Soil
If your soil has become compacted – likely the effect of heavy foot traffic – and is too dense for water, air and other nutrients to reach the roots of your grass, aerating can help break it up and reduce thatch. A core, or plug, aerator can introduce tiny holes into your soil by removing plugs of grass and soil, which lets nutrients more easily reach the roots. An added bonus, the plugs can decompose on top of your grass, supplying more nutrients.
Fill in Bare Spots
If your lawn is looking sparse, overseeding, which involves spreading grass seed over your existing lawn, can help fill in bare spots. Be sure to choose the right type of seed for your climate and soil type to ensure proper growth. Applying a slow-release nitrogen fertilizer at the same time can provide additional nutrients that are important for promoting healthy growth.
Start Seasonal Mowing
When the ground is dry enough and your grass is long enough to require cutting, begin seasonal mowing. Be sure to use proper techniques, including varying your mowing direction each time to avoid creating patterns or ruts, and avoid cutting grass too low, which can make the lawn more susceptible to weeds and drought stress. In general, never remove more than one-third of the grass blade at a time.
Find more tips to help get your yard ready for warm weather at eLivingtoday.com.
Photo courtesy of Unsplash
SOURCE:
eLivingtoday.com
Live Better
6 tips to improve sleep quality for overall well-being

(Family Features) Despite a rising number of people searching for the term “sleep” in 2023, nearly 1 in 3 U.S. adults report not getting enough of it, per the Centers for Disease Control and Prevention.
As a foundation for overall well-being, getting quality sleep is key to achieving goals and being present in daily life. Without it, you’re more likely to lack the stamina and energy to follow through on your ambitions.
To help people realize the foundational role sleep plays in everyday successes, Natrol – a leading sleep, mood and stress supplement brand based on Nielsen data – alongside Dr. Jess Andrade are working together to help people improve their sleep quality and habits so they can conquer their wellness goals this year and beyond.
“From consistent exercise to eating healthy and even daily journaling, it may seem like all your priorities are in order, but without sleep, you won’t be able to reap the full benefits of your hard work,” Andrade said. “Often overlooked, getting quality sleep is a fundamental piece to improving overall wellness and it doesn’t have to be complicated. Creating small, achievable changes in our daily sleep routines can lead to long-lasting lifestyle habits for the better.”
Consider these tips from Andrade to help improve your sleep quality so you can achieve your goals.
1. Prioritize Movement and Light Exposure in the Morning
Whether you’re a yogi or prefer a simple stretch when your feet hit the floor, movement and light during the day can help stimulate quality sleep later that night.
2. Understand Sleep Gains are Just as if Not More Important Than Gym Gains
If you’re focusing on fitness goals, you’ll need adequate sleep to see successful results. If you’re a morning workout warrior, schedule earlier bedtimes to ensure you get the recommended 7-8 hours of sleep. For evening workouts, aim to end your sweat session as early in the evening as possible; too much physical activity before bed can keep you up at night.
3. Make Your Bedroom a Sanctuary
Make your bedroom a sleep sanctuary. Keep the room dark, noise-free and comfortable with the thermostat set at 65-67 F for better sleep conditions.
4. Set Nightly Rituals to Unwind
Create nightly rituals that activate circadian rhythms and allow your body to unwind. Consider activities that relax you the most, like taking a bath, reading a book, journaling or sipping decaffeinated nighttime tea.
5. Try Incorporating a Drug-Free Sleep Aid
If you follow good bedtime habits but still occasionally struggle to fall or stay asleep, try a low-milligram melatonin supplement, like those from Natrol, that are designed to help you get a good night’s sleep so you wake up refreshed and ready to conquer your goals.†
6. Follow the 10-3-2-1-0 method
To help you fall asleep and wake up feeling revitalized, Andrade recommends the 10-3-2-1-0 method: Decrease caffeine intake at least 10 hours before bed. Avoid eating bothersome foods 3 hours prior to heading to off to sleep. Engage in relaxing activities like reading at the 2-hour mark and cut out screentime with 1 hour to spare. Ultimately, these habits can lead to zero times hitting snooze the next morning.
Find more ways to improve sleep habits by visiting Natrol.com.
† These statements have not been evaluated by the Food and Drug Administration. These products are not intended to diagnose, treat, cure, or prevent any disease.
SOURCE:
Natrol
Live Better
Find your perfect spring escape

(Family Features) Whether you’re a family with kids or a young professional looking for a getaway, it isn’t too late – or out of budget – to plan a memorable spring trip. All you need is a full tank of gas and your furry best friend to make an unforgettable getaway.
Data from online travel agency Booking.com shows that half of traveler’s plan to choose vacation destinations where the cost of living is less than their hometowns in 2024. Exploring lesser-known destinations with a variety of outdoor activities, opting for a road trip with your pet rather than a large group and traveling outside of peak season can all help make adventures more affordable.
To help travelers feel confident selecting their road trip destinations this spring, Motel 6, where pets always stay for free, and Bert Sperling’s Best Places recommend these undiscovered destinations that offer sight-seeing, access to dog parks, cultural experiences, green spaces, authentic cuisine, dog-friendly restaurants and affordable lodging.
Santa Fe, New Mexico
Dive into desert culture in Santa Fe with stunning views of the Sangre de Cristo mountains, Pueblo-style architecture, historic landmarks and pet-friendly dining patios. With near-endless activities like shopping for handcrafted jewelry, visiting the Museum of International Art Folk or walking the Santa Fe Plaza, there are entertainment options for everyone to enjoy. There are also plenty of affordable lodging options within walking distance of downtown attractions.
Branson, Missouri
Situated in the iconic Lake of the Ozarks, Branson is a dream small-town getaway for family vacations with a plethora of dining and entertainment options such as Silver Dollar City, Dolly Parton’s Stampede and the Titanic Museum. The city has a dog-friendly culture with plenty of parks and outdoor activities. Located just two miles from many of these local attractions, Motel 6 Branson welcomes the whole family, including those on four legs, at no additional cost. This location also offers amenities like free Wi-Fi, an expansive cable channel selection, a microwave and refrigerator in each room and guest laundry facilities.
Tempe, Arizona
If you’re seeking sunshine and fresh air, look no further than Tempe, a vibrant city located just south of Phoenix. From festivals and outdoor activities like golfing, hiking, kayaking or stand-up paddle boarding on Tempe Town Lake to visiting the Tempe Center for the Arts or local pet-friendly eateries, there are plenty of things to do in the low desert valley.
Chattanooga, Tennessee
For those looking to escape fast-paced city life with an outdoor getaway, Chattanooga is a perfect destination to enjoy outdoor activities, such as exploring Lookout Mountain or walking along the Tennessee River. In a city full of culture and history, visitors can enjoy local artwork at the Hunter Museum of American Art or go sightseeing in the historic Bluff View Art District. As the temperatures rise, embrace the rays at the seasonal pool alongside pet-friendly lodging at Motel 6 Chattanooga.
As you look to plan your getaway, visit Motel6.com to find pet-friendly and affordable lodging accommodations as well as more ideas to point you in the right direction on your spring excursion.
Photo courtesy of Shutterstock
SOURCE:
Motel 6
-

 NEWS1 year ago
NEWS1 year ago2 hurt, 1 jailed after shooting incident north of Nocona
-

 NEWS5 months ago
NEWS5 months agoSuspect indicted, jailed in Tia Hutson murder
-

 NEWS1 year ago
NEWS1 year agoSO investigating possible murder/suicide
-

 NEWS1 year ago
NEWS1 year agoWreck takes the life of BHS teen, 16
-
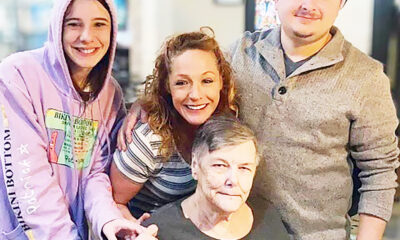
 NEWS9 months ago
NEWS9 months agoMurder unsolved – 1 year later Tia Hutson’s family angry, frustrated with no arrest
-

 NEWS12 months ago
NEWS12 months agoSheriff’s office called out to infant’s death
-

 NEWS1 year ago
NEWS1 year agoBowie Police face three-hour standoff after possible domestic fight
-

 NEWS1 year ago
NEWS1 year agoDriver stopped by a man running into the street, robbed at knifepoint





